CBD and THC Help Treat Neuroblastoma in Kids, Study Suggests
18 May, 2017
Findings in a new study show that cannabinoids impede the growth of tumors by reducing their viability and invasiveness, and inducing apoptosis.
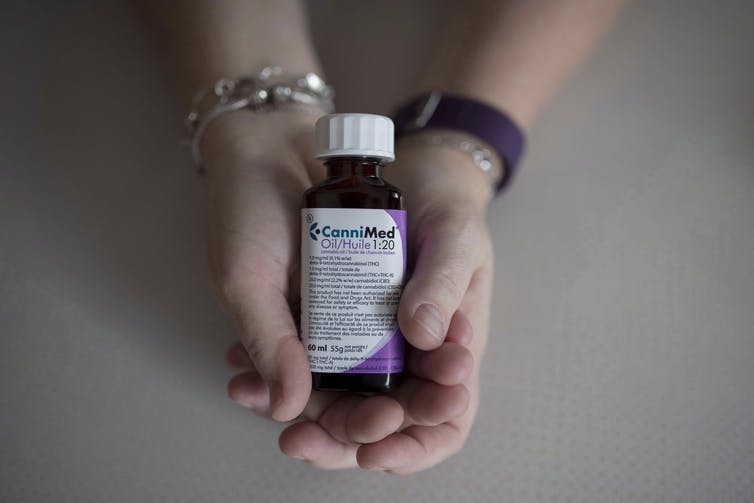
Israeli scientists have recently found that cannabinoids are effective at helping mitigate the rapid multiplication of neuroblastoma. In a new study, published in Current Oncology, researchers discovered that cannabidiol (CBD) and tetrahydrocannabinol (THC) show anti-tumor effects, while CBD was found to be the more active of the two.
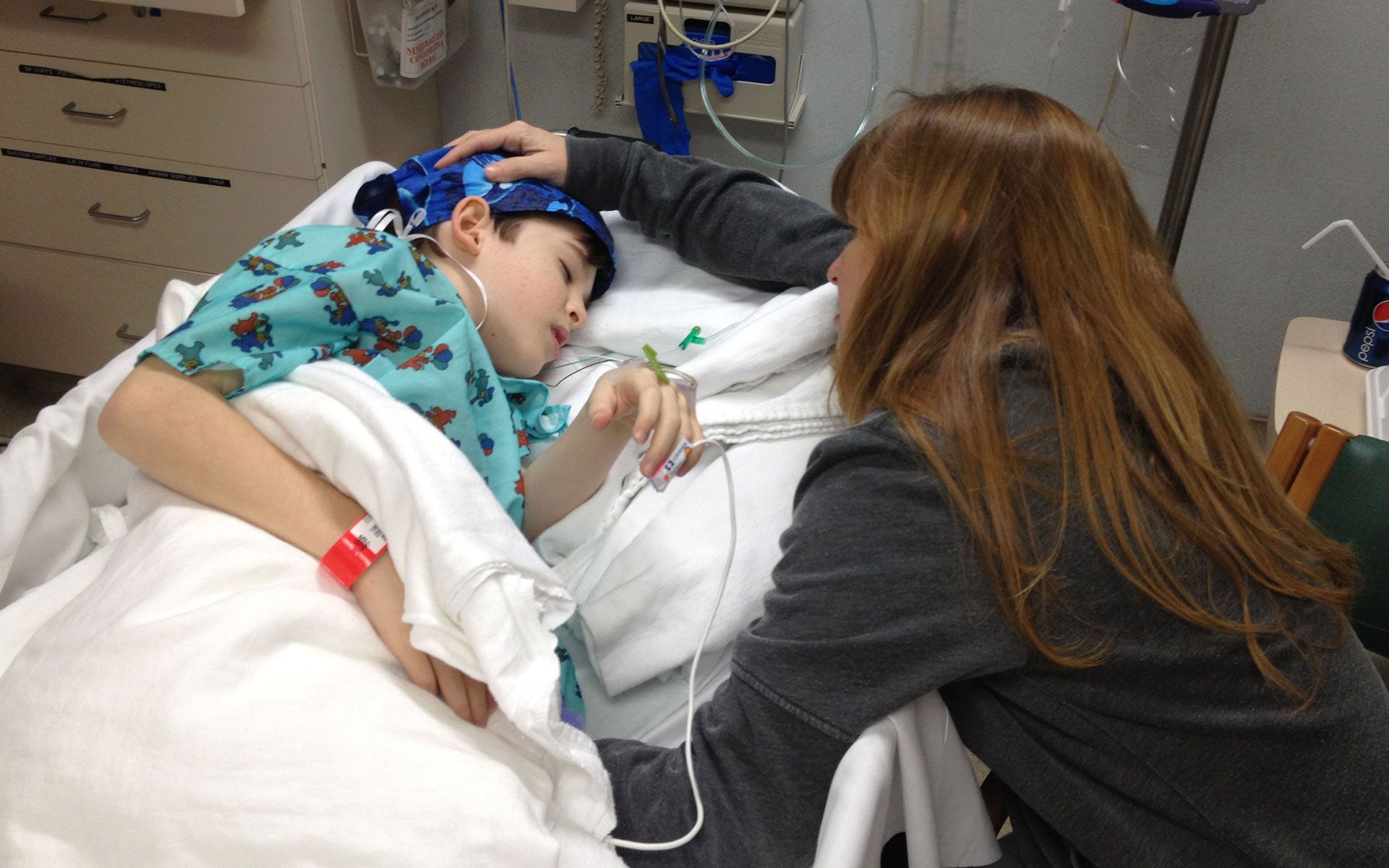
Neuroblastoma is a rare, but aggressive type of solid cancerous tumor. Affecting mostly infants and young children under five years of age, neuroblastoma begins in certain very early forms of nerve tissue found in an embryo or fetus. It’s most commonly found in the adrenal glands on top of the kidneys, but can also develop in the chest, belly, neck, pelvis, and in bones.
In the study, the researchers used culture and animal models to investigate the effects of CBD and THC, separately, on neuroblastoma. In the culture component, cells were treated with either of the cannabinoids or left untreated.
In the animal component of the study, tumors were induced in mice through subcutaneous injection. After allowing for the tumors to grow to an average size of 400 mm3, the researchers then injected the mice with either 20 mg/kg THC or 20 mg/kg CBD, or they left the mice untreated, for 14 days.
In both the culture cells and animal tests, the neuroblastoma tumors were measured and tested for viability, cell cycle distribution, growth rate, cell invasiveness, and apoptotic cell death rate.
Both compounds displayed anti-tumor activity by reducing tumor cell viability and effectively impeding tumor growth, although CBD’s effects were more significant. Additionally, treatment with CBD also reduced invasiveness of neuroblastoma cells and induced cancer cell death.
“Our findings about the activity of CBD in [neuroblastoma] support and extend previous findings about the anti-tumor activities of CBD in other tumors and suggest that cannabis extracts enriched in CBD and not in THC could be suitable for the development of novel non-psychotropic therapeutic strategies in [neuroblastoma],” the study concludes.
hempmeds-blog-image-what-is-cannabidiol-10-13-16-ver1
Previous preclinical studies have shown CBD to inhibit the progression of many types of cancer, including glioblastoma, breast, lung prostate, and colon cancers. The non-psychoactive cannabinoid has shown effective for inhibiting tumor cell viability, invasion, metastasis, and angiogenesis, as well as for inducing cancer cell death.
The researchers did not identify the mechanism for CBD’s anti-cancer effects, but noted that it appears independent of the cannabinoid’s influence on the endocannabinoid system’s CB1 and CB2 receptors.
While cannabis remains classified federally as a Schedule I substance – a category for substances that possess no therapeutic value — the U.S. National Cancer Institute has acknowledged the anti-cancer properties of cannabinoids like CBD. Because CBD is devoid of psychoactive effects, it may be a more suitable treatment option for cancers that affect children, such as neuroblastoma. In related news, investigators from Nottingham University’s world-leading Children’s Brain Tumor Research Centre recently announced a new study to investigate CBD’s effects on brain tumors in children.
You can read the entire study, “In vitro and in vivo efficacy of non-psychoactive cannabidiol in neuroblastoma,” via the National Center for Biotechnology Information, U.S. National Library of Medicine.
18 May, 2017
Findings in a new study show that cannabinoids impede the growth of tumors by reducing their viability and invasiveness, and inducing apoptosis.
Israeli scientists have recently found that cannabinoids are effective at helping mitigate the rapid multiplication of neuroblastoma. In a new study, published in Current Oncology, researchers discovered that cannabidiol (CBD) and tetrahydrocannabinol (THC) show anti-tumor effects, while CBD was found to be the more active of the two.
Neuroblastoma is a rare, but aggressive type of solid cancerous tumor. Affecting mostly infants and young children under five years of age, neuroblastoma begins in certain very early forms of nerve tissue found in an embryo or fetus. It’s most commonly found in the adrenal glands on top of the kidneys, but can also develop in the chest, belly, neck, pelvis, and in bones.
In the study, the researchers used culture and animal models to investigate the effects of CBD and THC, separately, on neuroblastoma. In the culture component, cells were treated with either of the cannabinoids or left untreated.
In the animal component of the study, tumors were induced in mice through subcutaneous injection. After allowing for the tumors to grow to an average size of 400 mm3, the researchers then injected the mice with either 20 mg/kg THC or 20 mg/kg CBD, or they left the mice untreated, for 14 days.
In both the culture cells and animal tests, the neuroblastoma tumors were measured and tested for viability, cell cycle distribution, growth rate, cell invasiveness, and apoptotic cell death rate.
Both compounds displayed anti-tumor activity by reducing tumor cell viability and effectively impeding tumor growth, although CBD’s effects were more significant. Additionally, treatment with CBD also reduced invasiveness of neuroblastoma cells and induced cancer cell death.
“Our findings about the activity of CBD in [neuroblastoma] support and extend previous findings about the anti-tumor activities of CBD in other tumors and suggest that cannabis extracts enriched in CBD and not in THC could be suitable for the development of novel non-psychotropic therapeutic strategies in [neuroblastoma],” the study concludes.
hempmeds-blog-image-what-is-cannabidiol-10-13-16-ver1
Previous preclinical studies have shown CBD to inhibit the progression of many types of cancer, including glioblastoma, breast, lung prostate, and colon cancers. The non-psychoactive cannabinoid has shown effective for inhibiting tumor cell viability, invasion, metastasis, and angiogenesis, as well as for inducing cancer cell death.
The researchers did not identify the mechanism for CBD’s anti-cancer effects, but noted that it appears independent of the cannabinoid’s influence on the endocannabinoid system’s CB1 and CB2 receptors.
While cannabis remains classified federally as a Schedule I substance – a category for substances that possess no therapeutic value — the U.S. National Cancer Institute has acknowledged the anti-cancer properties of cannabinoids like CBD. Because CBD is devoid of psychoactive effects, it may be a more suitable treatment option for cancers that affect children, such as neuroblastoma. In related news, investigators from Nottingham University’s world-leading Children’s Brain Tumor Research Centre recently announced a new study to investigate CBD’s effects on brain tumors in children.
You can read the entire study, “In vitro and in vivo efficacy of non-psychoactive cannabidiol in neuroblastoma,” via the National Center for Biotechnology Information, U.S. National Library of Medicine.